For more than 55 years, community health centers have provided comprehensive, culturally competent, high-quality primary care healthcare throughout the United States. Health centers are community-based and patient-directed organizations that deliver primary healthcare services to millions of people regardless of their ability to pay: “In 2021, health centers achieved a historic milestone of serving more than 30 million people . . . including one in three people living in poverty and one in five rural residents.”[1]
As designated by the Centers for Medicare and Medicaid Services, Federally Qualified Health Centers (FQHC) receive federal grants under section 330 of the Public Health Service Act (PHS Act) (42 U.S.C § 254b).[2]
Oversight of FQHCs is conducted by the Bureau of Primary Health Care (BPHC), which is one of the six branches of the Health Resources and Services Administration (HRSA) at the U.S. Department of Health and Human Services. “HRSA funds nearly 1,400 health centers and approximately 100 health center program look-alike organizations, collectively operating more than 14,000 service delivery sites in communities across the country.”[3]
Federal compliance requirements for FQHCs
In 2018, the BPHC first issued the Health Center Program Compliance Manual, which outlines the mandatory requirements for FQHCs. In developing the compliance manual, the BPHC produced a consolidated resource to assist health centers in understanding and demonstrating compliance with health center program requirements. The manual outlines federal requirements across its 21 chapters, which focus on:
-
Health center program eligibility
-
Health center program oversight
-
Needs assessment
-
Required and additional health services
-
Clinical staffing
-
Accessible locations and hours of operation
-
Coverage for medical emergencies during and after hours
-
Continuity of care
-
Sliding fee discount program
-
Quality improvement/assurance
-
Key management staff
-
Contracts and subawards
-
Conflicts of interest
-
Collaborative relationships
-
Financial management and accounting systems
-
Billing and collection
-
Budget
-
Program monitoring and data reporting systems
-
Board authority
-
Board composition
-
Federal Tort Claims Act (FTCA Redeeming Requirements)
The organization of each chapter addresses:
-
Authority, listing applicable statutory and regulatory citations.
-
Requirements, summarizing statutory and regulatory requirements.
-
Methods to display compliance, describing how FQHCs can demonstrate compliance with requirements.
-
Related considerations, explaining where FQHCs have discretion with respect to decision-making or which may be useful to consider when implementing a requirement.[4]
Federal mechanisms for assuring compliance with health center program requirements
The BPHC monitors FQHCs’ compliance with federal requirements through various mechanisms, including FQHCs’ timely submission of a series of mandatory reports and periodic on-site reviews—or operational site visit (OSV)—conducted at the health center by BPHC representatives.
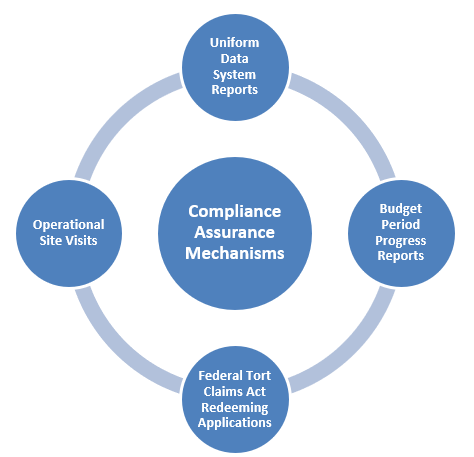
Federal reporting requirements for FQHCs include:
-
The Uniform Data System (UDS) report:
-
The annual UDS report, which covers the preceding calendar year, includes data on patient characteristics, services provided, clinical processes and health outcomes, patients’ use of services, staffing, costs, and revenues.[5]
-
-
The Budget Period Progress Report Non-Competing Continuation (BPR):
-
The BPR submission addresses the progress that a health center has made since its last application to HRSA, its expected progress for the remainder of the budget period and any projected changes.
-
-
The FTCA redeeming application:
-
On an annual basis, a redeeming application must be submitted to the bureau to maintain federal malpractice coverage through the FTCA for the health center, its clinicians, and clinical support staff in the next calendar year.
-
The on-site OSV provides an objective assessment and verification of the health center’s compliance with mandatory statutory and regulatory requirements. For health centers with a three-year project period, the OSV usually occurs within the project’s first 14–18 months. The OSV assesses an FQHC’s compliance with all program requirements outlined in Chapters 1 through 21 of the Health Center Program Compliance Manual.
Two chapters in the manual focus specifically on the governance of an FQHC by its board of directors (BOD): Chapter 19 addresses the board’s authority while Chapter 20 focuses on the board’s composition.



